
Centre for AIDS and Related Diseases
Officers
- Dr. Monil Singhai, Joint Director
- Dr. Preeti Patel, Assistant Director
Mandate of the Division
Centre for AIDS & Related Diseases (CA&RD) Division, NCDC, was established as the Apex Laboratory for HIV in 1985 and was subsequently renamed in 1995. The Centre was designated as one of the first National Reference Laboratory (NRL) for HIV by NACO in 2002. It achieved NABL accreditation in 2011 in accordance with ISO 15189 standards and has since maintained continuous accreditation, successfully transitioning to subsequent versions, including ISO 15189:2022.
The mandate of division is to support and strengthen national capacity for prevention, surveillance, and control of HIV and related diseases through quality diagnostics, technical expertise, and evidence-based public health support.
- The Centre provides HIV counseling and testing services to the walk-in clients, both provider-initiated and client-initiated.
- It is one of the 13 National Reference Laboratories (NRL) for HIV testing under the aegis of National AIDS Control Organization (NACO).
- The NRL is linked with 13 State Reference Laboratories (SRLs) in the states of Delhi, Haryana, Rajasthan and Jammu & Kashmir for External Quality Assessment Scheme (EQAS) for HIV testing and for confirmation of HIV serostatus of all samples with indeterminate / discordant resultsby Western Blot test.
- The NRL is a part of a Consortium of Kit Quality Testing Laboratories across India, with a mission to develop a national system for assuring Transfusion Transmitted Infections (TTI) test quality by a consortium with uniform approaches and procedures.
- The labs and units of the Centre participate in EQAS activities for HIV testing & CD4 count estimation and syphilis testing conducted by NARI Pune and VMMC &Safdarjung Hospital respectively.
- CARD functions as a NACO designated Referral Laboratory for HIV-2 confirmation
- Under HIV Sentinel Surveillance, the Centre is involved in HIV testing of Dried Blood Spot (DBS) samples and performing Quality Control testing of samples received from various Sentinel Sites in North India where the specimens are tested for four biomarkers i.e., HIV, Syphilis, HBV, and HCV.
- Testing of DBS samples under HIV Sentinel Surveillance for HIV, Syphilis, HBV and HCV.
- Absolute CD4 count and CD4 percentage estimation of HIV positive samples.
- Testing for Syphilis by RPR and TPHA.
- Capacity building activities at the Centre involve conducting EQAS Workshops for staff of linked SRLs, IEC activities for HIV/AIDS awareness.
- Provides technical support to the National AIDS Control Programme (NACP).
- CA&RD is one of the designated laboratories for HIV testing on DBS samples under National Family Health Survey-6 (NFHS-6).
Laboratories / Units under the Division
- Integrated Counseling and Testing Centre
- National Reference Laboratory for HIV
- Immunology Laboratory
- STI and Opportunistic Infections Laboratory
- HIV Serology Laboratory
- Blood Collection Centre
- HIV-2 Referral Laboratory
Achievements
- The Centre achieved NABL (National Accreditation Board for Testing and Calibration Laboratories) accreditation in the field of medical testing in the year 2011 and is obtaining its renewal from NABL every two years as per ISO 15189:2012.
- The National Reference Lab (NRL) at CARD is carrying out the preparation, maintenance and biannual distribution of Proficiency testing (PT) panels for HIV serology to 13 SRLs and to their linked 432 ICTCs in the Medical colleges in the states of Delhi, Rajasthan, Haryana and Jammu & Kashmir to ensure quality HIV testing.
- The NRL has successfully conducted workshops on EQAS for HIV Testing for Lab Technicians and Technical Officers of linked SRLs annually.
Staff Details
Head of the Division and Officers

Dr Monil Singhai
MBBS, MD
Joint Director
card@ncdc@gmail.com

Dr. Priti Patel
MBBS, MD
Assistant Director
card@ncdc@gmail.com

Mrs Sasmita Kar
B.Sc
Assistant Research Officer
sasmitakar@ncdc.gov.in

Mr Charan Singh
B.Sc
Assistant Research Officer
charansingh@ncdc.gov.in
Other Staff in the Division

Ms. Neha J. Tigga
M.Sc.
Research Assistant
nehatiggajy@gmail.com

Dr. Surbhi Mahajan
M.Sc., Ph.D
Research Assistant
surbhimahajan@gov.in

Ms. Preeti Diwakar
M.Sc.
Research Assistant
preetidiwakar1010@gmail.com

Ms. Hitakshi Setia
M.Sc., B.Ed.
Research Assistant
hitakshisetia@gmail.com

Mr. Ramchandra Bansfor
Intermediate
Technician

Mr. Divakar Sharma
B.Sc.
Technician
divakarsharma28899@gmail.com

Mr. Rajesh Kumar
HSC
Laboratory Assistant
rajeshncdc71@gmail.com

Mr. Mohit Kumar
Diploma
MTS
mohitkumarkumar191@gmail.com

Mamta Sungroya
B.Sc, B.Ed
Lab Attendant

Ankur Verma
12th
Lab Attendant
Relevant photographs of various activities of the CA&RD division –
Referral Diagnostic Services
- Act as a national referral diagnostic services provider for HIV and related diseases, including confirmation of complex, indeterminate, and discordant test results, and support to Linked State Reference Laboratories and ICTCs.
Proficiency Testing (PT) Provider
- Serve as a Proficiency Testing provider for HIV, ensuring quality assurance, standardization, and capacity strengthening of linked laboratories.
Capacity Building and Training
Plan and conduct workshops, trainings, and programmes on:
- Biosafety and biosecurity
- Good Laboratory Practices (GLP)
- Quality Management Systems
- Diagnostic technologies
Expert Representation at National Level
-
Provide technical expertise in committees:
- National AIDS Control Organisation (NACO)
- Bureau of Indian Standards (BIS)
- Drugs Controller General of India (DCGI)
- National One Health Mission (NOHM)
- Contribute to policy formulation and guideline development.

Good Laboratory Practices Refresher and flow cytometry Training for Lab Technicians for 7 states (19-22.8.2025)

External Quality Assurance Scheme Workshop for laboratory technicians for 13 states (1.12.2025)


HIV-awareness programme on World AIDS Day (1.12.2025)


Participated in Workshop on NFHS-6
IEC activities for HIV/AIDS awareness –



The Centre has successfully organized a Nukkad Natak (street play) to raise awareness on HIV/AIDS on the occasion of World AIDS Day on 01st December 2022 for all staff and officers of NCDC and NCVBDC along with daily wage workers and construction workers who are presently working in NCDC campus.
Poster Making Competition on the eve of World AIDS Day

NCDC conducts Workshop on “External Quality Assessment Scheme (EQAS) for HIV testing” for 13 State Reference Laboratories (SRLs)



Hands-on Training on ELISA technique of technical staff at NCDC, Delhi

Training of technical staff from various laboratory divisions on Bio-medical Waste Management at Central Bio-medical Waste Storage Facility at NCDC, Delhi
Publications
- Agarwal, S. K., Singh, A., Anuradha, S., Singh, N. P., Sokhi, J., & Baveja, U. K. (2001). Cytokine profile in human immunodeficiency virus positive patients with and without tuberculosis. The Journal of the Association of Physicians of India, 49, 799–802.
- Bajaj, J. S., Singh, A., Aggarwal, S. K., Chattopadhya, D., & Baveja, U. K. (2000). Synergistic immunosuppression by candida in HIV infection: a cytokine based analysis. The Journal of Communicable Diseases, 32(1), 1–9.
- Baveja, U. K., Chattopadhya, D., Khera, R., & Joshi, P. M. (2003). A cross sectional serological study of the co-infection of hepatitis B virus, hepatitis C virus and human immunodeficiency virus amongst a cohort of IDUs at Delhi. Indian Journal of Medical Microbiology, 21(4), 280–283.
- Baweja, U. K., Talukdar, P. K., & Singh, R. (1986). A clinicoparasitological study of scabies. The Journal of Communicable Diseases, 18(2), 132–135.
- Chattopadhya, D., Aggarwal, R. K., Baveja, U. K., Doda, V., & Kumari, S. (1998). Evaluation of epidemiological and serological predictors of human immunodeficiency virus type-1 (HIV-1) infection among high risk professional blood donors with western blot indeterminate results. Journal of Clinical Virology: The Official Publication of the Pan American Society for Clinical Virology, 11(1), 39–49 https://doi.org/10.1016/s0928-0197(98)00041-5
- Chattopadhya, D., Baveja, U. K., Bose, M., & Kumar, A. (2002). Disease progression markers during asymptomatic phase of HIV-1 infected children with unimpaired CD4+ cell values: evaluation of repeat CD4+ cell evaluation vs. other immunological parameters. Journal of Tropical Pediatrics, 48(6), 340–347. https://doi.org/10.1093/tropej/48.6.340
- Chattopadhya, D., Grover, S. S., Sharma, M., Ichhpujani, R. L., & Baveja, U. K. (2003). Immune response in HIV-1-infected children with thalassaemia given a primary course of DPT vaccine before acquiring HIV-1 infection. Annals of Tropical Paediatrics, 23(4), 279–292. https://doi.org/10.1179/027249303225007824
- Dwivedi, K. K., Prasad, G., Saini, S., Mahajan, S., Lal, S., & Baveja, U. K. (2007). Enteric opportunistic parasites among HIV infected individuals: associated risk factors and immune status. Japanese Journal of Infectious Diseases, 60(2–3), 76–81. https://doi.org/10.7883/yoken.jjid.2007.76
- Gugnani, H. C., Becker, K., Fegeler, W., Basu, S., Chattopadhya, D., Baveja, U., Satyanarayana, S., Kalghatgi, T., & Murlidhar, A. (2003). Oropharyngeal carriage of Candida species in HIV-infected patients in India. Mycoses, 46(8), 299–306. https://doi.org/10.1046/j.1439-0507.2003.00896.x
- Hussain, Z., Das, B. C., Husain, S. A., Polipalli, S. K., Ahmed, T., Begum, N., Medhi, S., Verghese, A., Raish, M., Theamboonlers, A., Poovorawan, Y., & Kar, P. (2006). Virological course of hepatitis A virus as determined by real time RT-PCR: Correlation with biochemical, immunological and genotypic profiles. World Journal of Gastroenterology, 12(29), 4683–4688. https://doi.org/10.3748/wjg.v12.i29.4683
- Kaur, R., Berry, N., Mittal, S. K., Mathur, M. D., & Baveja, U. (2002). Hepatitis B virus in a select pediatric population in Delhi, India. The Journal of Communicable Diseases, 34(2), 146–148.
- Kaur, R., Mittal, N., Bhalla, P., Reddy, B. N. S., & Baveja, U. K. (2006). Risk factors of herpes simplex virus type 2 among STD clinic attenders in Delhi, India. The Journal of Communicable Diseases, 38(4), 339–343.
- Sehgal, R., Baveja, U. K., Chattopadhya, D., Chandra, J., & Lal, S. (2005). Pediatric HIV infection. Indian Journal of Pediatrics, 72(11), 925–930. https://doi.org/10.1007/BF02731665
- Shivraj, S. O., Chattopadhya, D., Grover, G., Kumar, A., & Baveja, U. K. (2006). Role of HCV coinfection towards disease progression and survival in HIV-1 infected children: a follow-up study of 10 years. Journal of Tropical Pediatrics, 52(3), 206–211. https://doi.org/10.1093/tropej/fmi103
Technical Guidelines
-
Biosafety manual for public health laboratories
View Document -
National Strategic Plan for HIV/AIDS and STI (2017–2024)
View Document -
Consolidating the Evidence, Building the Future: Report of Consultative Meeting on Integrated and Enhanced Epidemiology under NACP
View Document

Integrated Counselling and Testing Services
- Counseling for HIV/AIDS and related Diseases: Pre and post test counseling of walk-in clients attending Integrated and counseling and testing centre (ICTC).
- All confirmed HIV positive patients tested at ICTC are referred to Antiretroviral Therapy (ART) centers for treatment.
- All Syphilis positive patients are referred to STI centers for treatment and all suspected TB patients are referred to RNTCP.
- Provide services of Awareness on HIV/AIDS and facilitation of HIV Counseling & Testing of general population and high-risk population in different regions of Delhi.
National Reference Laboratory for HIV
2.1 Diagnostic Laboratory
The aim is to provide confirmation of HIV and Related diseases is done for walk-in, referred and HIV indeterminate samples from linked SRLs and ICTCs
Name of tests done with turnaround time
| Name of Test | Turnaround Time |
|---|---|
| HIV Rapid Test (ICTC Client) | One working day |
| HIV ELISA Test (Referred) | Seven working days |
| HIV-1/2 Western blot (Referred) | One Month |
| CD4 Count Test (Referred) | One working day |
| Serological test for Syphilis test (Walk-in/Referred) (Rapid Test) | Two working days |
| Serological test for Syphilis test (Walk-in/Referred) (TPHA Test) | Two working days |
2.2 Proficiency testing Panel Preparation Laboratory
Preparation and distribution of proficiency testing (PT) panel of HIV for linked SRLs and their associated ICTCs
| Disease | Sample | Panel Size | Test |
|---|---|---|---|
| HIV-1 / HIV-2 | Serum / Plasma / Whole Blood | State Reference Laboratories-8 members panel ICTCs-4 members panel |
RDT, ELISA |
States covered:4 {Delhi (4), Haryana (1), Jammu & Kashmir (2), Rajasthan (6)}
SRLs:13
ICTCs:400
Frequency: Twice a year
2.3 HIV and related diseases Surveillance Laboratory
1. a) National Family Health Survey (NFHS)
Laboratory support to generate reliable national prevalence estimates by providing technical expertise, laboratory support, and quality assurance for HIV testing as and when required
1. b) Interlaboratory Comparison/Retesting
| Sample | Disease | Test |
|---|---|---|
| Serum | HIV, HBV, HCV | RDT, ELISA |
| Serum | Syphilis | RDT, RPR, TPHA |
| Dried Blood Spot | HIV | ELISA |
2.4 Kit Evaluation Laboratory
| Test | Principle | Panel Size |
|---|---|---|
| HIV | Immunochromatography/ Immunodot | ~500 (100 positive + 400 Negative) |
| HBV | Immunochromatography | ~500 (100 positive + 400 Negative) |
| HCV | Immunochromatography | ~500 (100 positive + 400 Negative) |
| HIV + Syphilis (Dual kit) | Immunochromatography | ~575 (100 positive + 400 Negative) |
Services Provided by CA&RD Division
- Counseling for HIV/AIDS: Pre and post test counseling of walk-in clients attending Integrated and counseling and testing centre (ICTC).
- All confirmed HIV positive patients tested at ICTC are referred to Antiretroviral Therapy (ART) centres for treatment. All Syphilis positive patients are referred to STI centres for treatment and all suspected TB patients are referred to RNTCP.
- Provide services of Awareness on HIV/AIDS and facilitation of HIV Counseling & Testing of general population and high risk population in different regions of Delhi.
- HIV-2 referral centre: Confirmation of HIV-1+2 and HIV-2 is done for HIV positive patients referred from ART centres of Delhi, Haryana, Rajasthan, Punjab, Jammu & Kashmir, Himachal Pradesh and Chandigarh.
- Evaluation of diagnostic kits (Rapid and ELISA) for HIV/HBV/HCV.
Name of tests done with turnaround time
| Name of tests | Turnaround time |
|---|---|
| HIV test (ICTC Client) | One working Day |
| HIV test (Referred) | ELISA-: 7 Working days Western blot : One month |
| CD4 count test (Referred HIV Positive samples from ART centres) | One working day |
| Syphilis test | Two working days |
| Kit evaluation of HIV/ HBV/ HCV | 10 working days |
Name and contact number of concerned official/officer-
ICTC Counselor:011-23909327

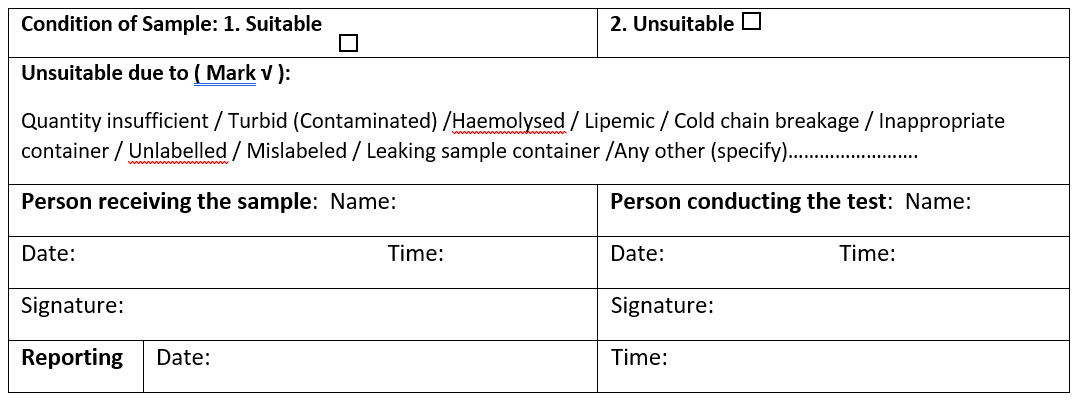
PART-II: PRIMARY SAMPLE COLLECTION
(To be filled by CARD Blood Collection Centre / Linked Centers collecting blood)

PART-III: SUITABILITY OF SAMPLE
(To be filled by ICTC / Serology / Immunology Laboratory/ STD-OI Laboratory)
Contact Us
Full Mailing Address:
Centre for AIDS and Related Diseases (CA&RD)
National Centre for Disease Control
Ministry of Health and Family Welfare
Address Details: N/A
Phone: 011-20832460
Email: card.ncdc@gmail.com


